
Top 10 Raw Pharmaceutical Ingredients You Should Know?
In the intricate world of pharmaceuticals, one aspect stands out: Raw Pharmaceutical ingredients. These essential components form the backbone of many medications. Understanding their significance is crucial for both professionals and consumers. This knowledge helps ensure safety and efficacy in drug production.
The selection of quality raw pharmaceutical ingredients directly impacts the overall effectiveness of treatments. There is an array of ingredients, each serving unique roles in formulations. However, not all raw pharmaceutical materials are equal. Some may pose risks if not sourced properly. Consumers must remain vigilant and informed.
Expertise in identifying reputable suppliers of raw pharmaceutical ingredients is essential. Knowledge of these components fosters trust and transparency in the pharmaceutical industry. While navigating this complex landscape, it’s vital to consider both the benefits and the potential pitfalls. Awareness of these aspects can lead to better health outcomes and smarter choices.

Understanding Raw Pharmaceutical Ingredients and Their Importance
In the pharmaceutical industry, raw ingredients form the backbone of drug development. Understanding these components is crucial for manufacturers. Each ingredient affects the drug’s efficacy and safety. Quality control is, therefore, paramount. A single impurity can derail the healing process and compromise patient safety.
Choosing the right raw materials involves careful consideration. Look for suppliers with a proven track record. Ask about their quality assurance practices. Reliable suppliers often provide documentation to ensure compliance with industry standards. Connections in the industry can offer valuable insights as well.
Keep in mind that not all raw ingredients are created equal. Some may have varying levels of purity. Scrutinizing them is essential. Testing different samples can help in selecting the best option. Regular assessments of ingredient quality are necessary. This not only boosts credibility but also fosters trust with end-users. Exploring partnerships with reputable laboratories can enhance your understanding of these materials.
Top 10 Raw Pharmaceutical Ingredients
This bar chart illustrates the significance of various raw pharmaceutical ingredients based on their usage in drug formulation.
The Role of Active Pharmaceutical Ingredients (APIs) in Drug Development
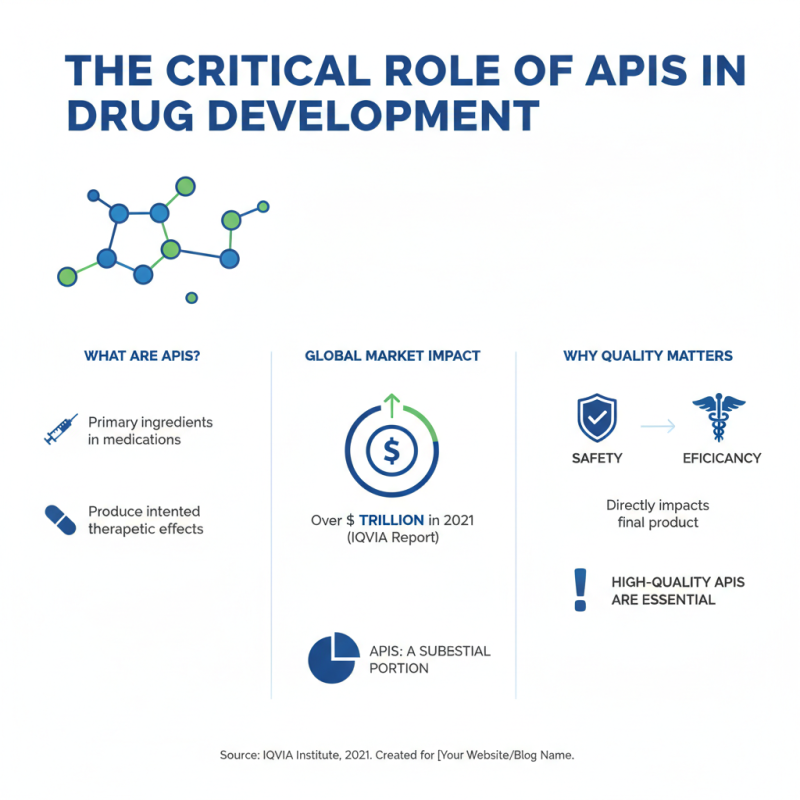
Active Pharmaceutical Ingredients (APIs) play a critical role in drug development. They are the primary elements that produce the intended therapeutic effects in medications. According to a report by the IQVIA Institute, the global pharmaceutical market was valued at over $1 trillion in 2021, with APIs representing a substantial portion of this market. The importance of high-quality APIs cannot be overstated, as they directly impact the safety and efficacy of the final product.
The sourcing and manufacturing of APIs are complex processes. Approximately 70% of APIs are produced in Asia, particularly in China and India. This geographic concentration raises concerns about supply chain dependencies and quality control. A significant number of drug recalls have been attributed to API quality issues. These challenges highlight the ongoing need for stringent regulatory oversight. The U.S. FDA and EMA continually enhance their guidelines, but maintaining consistent quality remains a significant hurdle.
Recent trends indicate an increased push towards innovation in API production. Technologies like continuous manufacturing and biotechnological processes are emerging to improve efficiency. However, these advancements also require skilled personnel and significant investment. Many companies face challenges in adopting these new technologies, reflecting a broader industry concern. Balancing innovation with regulatory compliance and cost-effectiveness is an ongoing struggle for pharmaceutical firms.
Commonly Used Excipients and Their Functions in Formulations
Excipients play a crucial role in the pharmaceutical industry. They enhance the stability and delivery of active ingredients. Common excipients include fillers, binders, and preservatives. According to a report by Grand View Research, the global excipient market is projected to reach $9.5 billion by 2026, emphasizing their importance.
Some excipients serve specific purposes. For instance, lactose is often used as a filler. It helps improve the bulk of tablets. Starches act as binders, ensuring the proper formulation of pills. Preservatives like sodium benzoate help maintain product shelf life.
**Tip:** Always check for potential allergens in excipients. They can affect sensitive individuals.
The landscape of excipients is evolving. Newer alternatives are gaining recognition. Natural excipients are becoming popular due to their safety profiles. However, the selection process is not always straightforward.
**Tip:** Conduct thorough research about excipient sources. Ensure they meet regulatory standards and quality control measures.
Top Ten Raw Pharmaceutical Ingredients and Their Key Characteristics
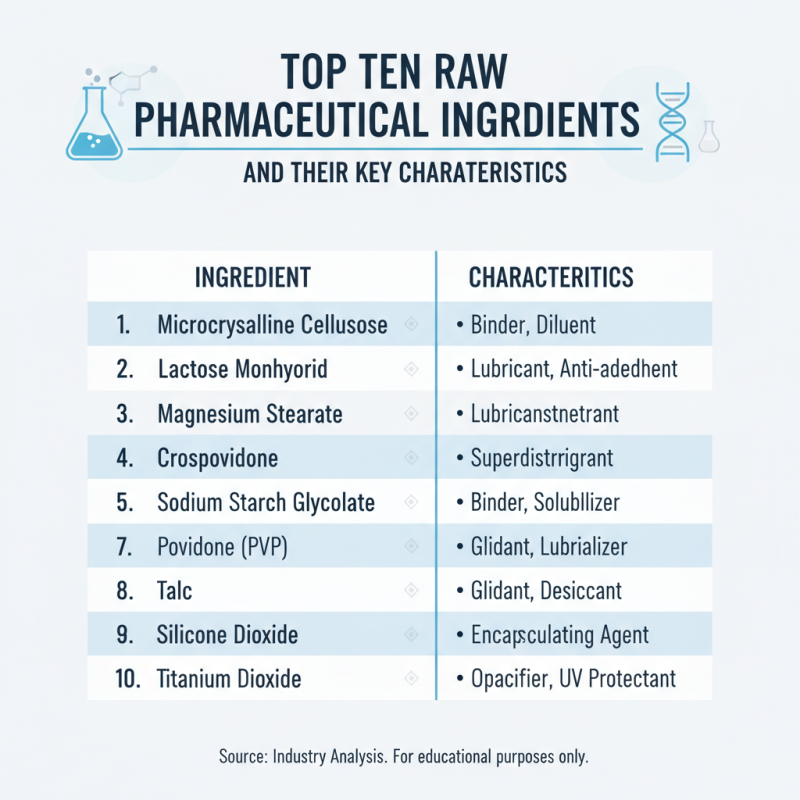
Understanding raw pharmaceutical ingredients is crucial for anyone in the industry. These components form the basis of drug manufacturing. Here are ten key ingredients along with their primary characteristics.
Active Pharmaceutical Ingredients (APIs) stand at the forefront. They are essential for therapeutic effects. Over 90% of all drugs rely on highly pure APIs for effectiveness. Another critical component is excipients. They help with drug stability and absorption. Their role is often underestimated, yet they make up to 50% of a formulation.
A notable ingredient is solvents. They dissolve other substances, making formulations easier to handle. Many solvents are recognized for their biocompatibility, ensuring product safety. APIs often come from various sources, including synthetic and natural origins. The choice impacts the drug's efficacy and production costs.
**Tips:** Always analyze the source of APIs. Their origin can significantly affect quality. Ensure excipients are selected based on compatibility with the API. A mismatch can lead to serious formulation issues. Continuously update your knowledge on regulatory changes affecting ingredients.
Adopting a rigorous evaluation process is essential. However, it's important to be aware of potential supply chain disruptions. Relying heavily on specific ingredients can create vulnerabilities. The industry must focus on diversifying sources for better resilience and stability.
Regulatory Standards and Quality Control in Pharmaceutical Manufacturing
In pharmaceutical manufacturing, adhering to regulatory standards is critical. The FDA and EMA set strict guidelines to ensure safety and efficacy. According to a recent report by PharmTech, over 70% of quality issues arise from raw ingredients. These deficiencies can lead to product recalls and financial losses.
Quality control is essential in sourcing raw materials. Manufacturers should validate suppliers rigorously. Batch testing and verification help to maintain ingredient integrity. Compliance with Good Manufacturing Practices (GMP) is non-negotiable. A survey indicated that companies with robust quality systems see 30% fewer quality issues.
Tip: Regularly audit suppliers to assess adherence to quality standards.
Be aware that not all suppliers meet these stringent criteria. Inconsistent quality can compromise drug effectiveness. It’s vital to embrace a culture of quality throughout your organization. Small lapses in quality can lead to significant consequences, affecting patient safety and brand reputation.
Tip: Invest in training your team on regulatory changes and quality management.
Related Posts
-
What Are the Uses and Benefits of Pharmaceutical Chemical Powder?
-

Top 10 Pharmaceutical Chemicals in China You Should Know?
-

Essential Tips for Sourcing Pharmaceutical Intermediates?
-
5 Essential Tips for Sourcing Quality Pharmaceutical Materials
-

Navigating the Future of Best Pharmaceutical Materials in 2025 and How to Stay Ahead
-

How to Optimize Your Supply Chain for Drug Raw Material Sourcing and Compliance
