
Ultimate Tips for Choosing the Best Drug Capsule Material?
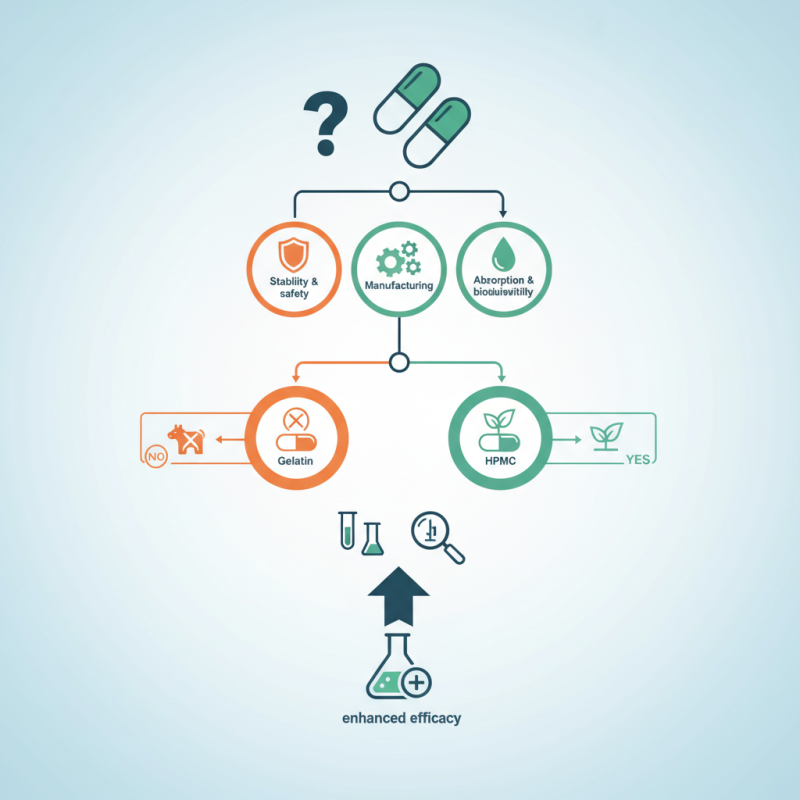
Choosing the right drug capsule material is crucial for pharmaceutical development. This decision impacts absorption, bioavailability, and patient compliance. Factors such as stability, safety, and manufacturing processes must be carefully evaluated.
Different materials, like gelatin and HPMC, each have unique properties. Gelatin capsules are versatile and widely used. However, they may not be suitable for vegan products. HPMC capsules, on the other hand, offer a plant-based alternative. This flexibility is vital in an ever-diversifying market.
Understanding the specific needs of your formulation is essential. Certain drug types may react poorly with certain capsule materials. Prior knowledge can guide this choice. Not all materials are created equal, and assessing quality is fundamental. Consider conducting thorough testing to ensure reliability. Making an informed decision will enhance the efficacy of your product.
Understanding Different Types of Drug Capsule Materials Available
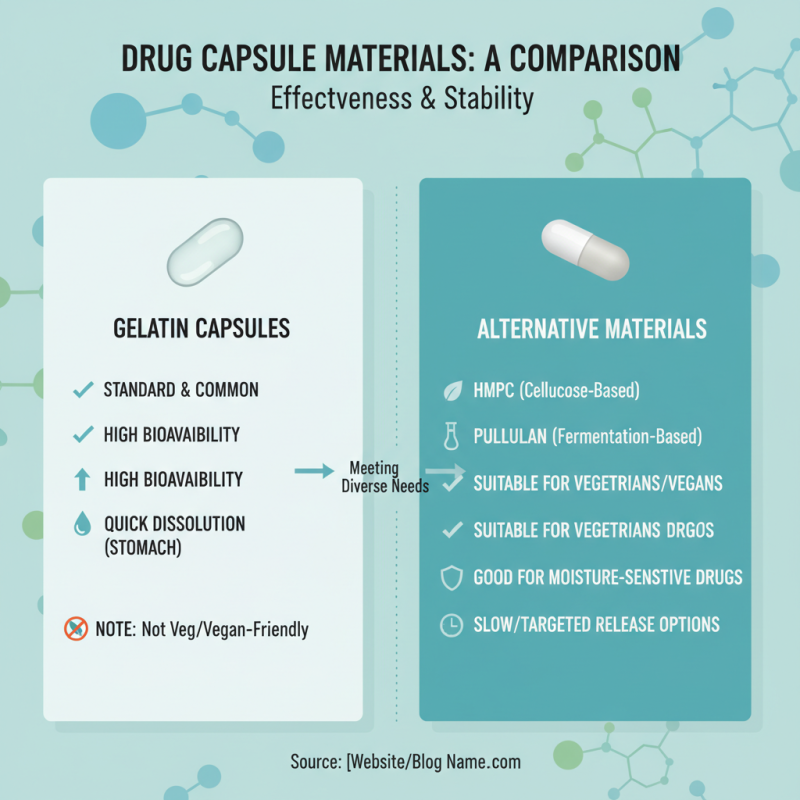
The choice of drug capsule material significantly impacts the effectiveness and stability of medications. Different materials provide unique advantages and challenges. Gelatin capsules remain a standard due to their bioavailability and ability to dissolve quickly in the stomach. However, they may not be suitable for vegetarians or certain dietary restrictions, drawing attention to alternatives.
Vegetable cellulose capsules have gained popularity, particularly with the rise of dietary supplements. Studies show that over 30% of consumers prefer plant-based options. These capsules offer comparable dissolution rates to gelatin types. However, one must consider their potential moisture sensitivity. An incorrect storage environment could compromise the capsule's integrity.
Polyelectrolyte-based materials also show promise in controlled drug release applications. They can adjust the dissolution rate based on environmental conditions. Yet, more research is necessary to optimize their use in mainstream pharmaceuticals. It is essential to weigh these materials' benefits against their limitations, ensuring the choice aligns with specific therapeutic needs and patient preferences.
Evaluating Factors Affecting the Selection of Capsule Materials
Choosing the best drug capsule material involves careful consideration of various factors. The selection process should prioritize the stability of the drug substance. Factors like moisture sensitivity and temperature fluctuations can lead to degradation. A 2021 report from the International Pharmaceutical Federation highlights that over 20% of drug failures are attributed to inadequate capsule protection.
Another critical aspect is the compatibility of the capsule material with the active pharmaceutical ingredient (API). Different materials may react differently to various APIs. For instance, gelatin capsules might not be suitable for hygroscopic substances. According to the World Health Organization, over 30% of patients prefer capsules for their ease of swallowing, emphasizing their importance in drug delivery.
Tips for selecting capsule materials include conducting stability studies early on. This can reveal the potential for interactions between the capsule and the drug. Another recommendation is to consider the manufacturing process. If high temperatures are used, the material’s thermal stability becomes vital. Regularly reviewing industry guidelines can ensure adherence to best practices, fostering reliability in capsule selection. Balancing these elements can lead to effective formulation strategies.
Comparing Gelatin and Vegetarian Capsule Options
When it comes to choosing capsule materials, the debate between gelatin and vegetarian options is essential. Gelatin capsules are derived from animal collagen. They offer a seamless dissolution in the stomach. Their texture can provide a familiar experience for users. However, these capsules may not align with vegan or vegetarian lifestyles. This can create a gap for consumers who prioritize their dietary choices.
Vegetarian capsules, often made from hydroxypropyl methylcellulose (HPMC), present an appealing alternative. They cater to those with ethical or dietary constraints. These capsules can also provide excellent stability and a neutral taste. While they are suitable for a broader audience, some users may find them less familiar. Moreover, the production process often requires scrutiny to ensure quality.
Both options have their pros and cons. Some consumers may prefer gelatin for its traditional appeal. Others may wish to explore plant-based options. Each choice leads to unique considerations. It’s essential to assess the target consumer base and their preferences. Understanding these factors is crucial for making the best decision.
Ultimate Tips for Choosing the Best Drug Capsule Material - Comparing Gelatin and Vegetarian Capsule Options
| Feature | Gelatin Capsules | Vegetarian Capsules |
|---|---|---|
| Source Material | Animal bones or skin | Plant cellulose |
| Gelatin Type | Hydrolyzed gelatin | Pullulan or hydroxypropyl methylcellulose (HPMC) |
| Vegan Friendly | No | Yes |
| Dissolution Time | Faster (around 15 minutes) | Moderate (up to 30 minutes) |
| Temperature Sensitivity | Sensitive to heat | More stable at varying temperatures |
| Cost | Generally lower | Typically higher |
| Taste Masking | Good | Better for strong tastes |
Assessing Capsule Material Stability and Drug Compatibility
When choosing drug capsule materials, drug compatibility and stability are crucial factors to consider. The material must not react adversely with the active ingredients. This reaction can compromise the drug's efficacy. For instance, some gelatin capsules can interact with moisture, leading to degradation of the content.
Stability also involves temperature and humidity considerations. Some materials may escape their intended storage conditions, leading to unexpected results. It’s essential to conduct thorough stability testing for specific drugs. Manufacturers should assess how the capsule behaves under different conditions. This assessment aids in ensuring the drug maintains its potency throughout its shelf life.
It's worth noting that not all materials are universally compatible. Certain coatings might limit the release of the drug, impacting absorption. This limitation necessitates fine-tuning the formulation process. Continuous feedback and studies are essential in enhancing the selection of capsule materials. The quest for the perfect capsule is ongoing, indicating a need for constant evaluation in the pharmaceutical field.
Regulatory Considerations for Capsule Material Choices
When selecting capsule materials, regulatory considerations play a pivotal role. Compliance with local and international regulations is essential. Different countries have varying standards for excipients and additives. Understanding these regulations ensures that the chosen materials are safe and effective. It reduces the risk of recalls or legal issues.
Capsule materials must also be compatible with the drug formulation. Some substances may react negatively, compromising the drug's integrity. Evaluating stability under specific storage conditions is crucial. Testing for interaction between the capsule and the active ingredients can help ensure safety.
Here are some tips for selecting capsule materials:
- Evaluate the drug's properties. Consider factors like solubility and release profile.
- Consult with regulatory bodies early in the development process. This can guide compliant choices.
- Review relevant pharmacopoeias. They provide standards for capsule materials, ensuring safety and efficacy.
Awareness of these considerations helps manufacturers avoid costly mistakes. Regular audits and updates on regulations can further enhance compliance and trust. It's a complex landscape, but informed choices lead to better products.
Related Posts
-

Essential Guide to Sourcing Pharma Materials for Global Buyers
-

Solutions for Sourcing the Best Pharma Materials in the Industry
-

How to Effectively Source and Manage Pharma Material for Your Business Success
-

Top Tips for Choosing the Right Medicine Capsule Material for Your Needs
-

Unlocking the Future of Biopharmaceuticals with Peptide Materials in Global Supply Chains
-

2026 Top Pharmaceutical Supplier Trends and Innovations Revealed?
