
Pharma Material Tips for Effective Management and Usage?
Effective management and usage of Pharma Material is crucial in the pharmaceutical industry. Handling these materials requires precision and care. Poor management can lead to wastage, delays, and compromised product quality. This impacts not just costs but also patient safety.
Pharma Material involves various substances, from active ingredients to packaging components. Each type has specific storage and handling needs. Many professionals overlook these details. They often assume that all materials can be treated similarly. This can result in severe consequences, including contamination.
It is vital to develop good practices for managing Pharma Material. Regular training and updates on protocols can ensure better outcomes. People must reflect on their methods and make improvements. Small changes can have significant effects. Engaging with suppliers and understanding material specifications helps too.

Pharmaceutical Material Overview: Types and Classifications in the Industry
In the pharmaceutical industry, materials are crucial for effective product development. These materials can be classified into several categories. Active Pharmaceutical Ingredients (APIs) are the core components that provide therapeutic effects. Excipients, on the other hand, serve various functions, including stabilizing the formulation and enhancing drug delivery.
Another important category is packaging materials. These ensure the product's integrity and provide crucial labeling information. Quality control is vital here. Poor material selection can lead to failures during production. It may seem trivial, but the wrong excipient can alter the drug’s efficacy.
In the rush to innovate, there’s a tendency to overlook details. Many managers underestimate the importance of material sourcing. Cost-cutting measures can backfire, resulting in subpar products. Every supplier should be thoroughly vetted. What seems like a reasonable choice today may lead to compliance issues tomorrow. Reflecting on these choices is essential for long-term success.
Key Metrics for Evaluating Pharma Materials: Quality and Compliance Standards
Evaluating pharma materials is crucial for effective management. Quality is a primary metric. It ensures that materials meet necessary specifications. Compliance standards must be adhered to strictly. This includes regulatory requirements and industry best practices. Meeting these standards can often be a challenge. Variability in raw material quality can affect outcomes.
Organizations sometimes overlook the importance of thorough testing. Quality assurance processes should be robust but can be costly. Regular audits are essential, yet some may find them tedious. The complexity of supply chains adds another layer. Tracking compliance across various suppliers can become overwhelming.
Effective management of pharma materials requires ongoing reflection. Continuous improvement should be the goal. Metrics should be revisited regularly to assess relevance and effectiveness. This iterative process helps identify weaknesses and opportunities. A commitment to quality and compliance can protect patient safety and enhance credibility in the industry.
Best Practices for Inventory Management in Pharmaceutical Materials
Effective inventory management is crucial in the pharmaceutical industry. A recent report indicates that over 30% of pharmaceutical companies struggle with issues related to inventory discrepancies. Incorrect inventory can lead to costly production delays and compliance issues. Maintaining accurate stock levels is essential.
Regular audits and real-time tracking can help address discrepancies. Tip: Implement a barcode system to enhance tracking accuracy. This method has shown to reduce inventory errors by up to 25%. It's vital to ensure that all team members are trained in using this system effectively.
Another common challenge is managing expiry dates. With many materials having short shelf lives, it's easy for items to become obsolete. Tip: Utilize a first-expiry, first-out (FEFO) system. This can improve turnover rates and reduce waste. Despite these strategies, some companies still find themselves facing challenges in keeping materials updated.
Adapting to new technologies can enhance inventory efficiency. However, not every organization is fully utilizing these tools. Continuous training and assessment are necessary for effective management.
Strategies for Cost-Effective Usage of Pharma Materials in Production

In the pharmaceutical industry, managing materials efficiently is crucial for cost-effectiveness. One strategy involves careful inventory management. Keeping track of stock levels can prevent excess waste. Outdated materials can lead to losses, so regular audits are essential. Streamlining the supply chain also plays a significant role. Establishing strong relationships with suppliers can reduce costs.
Another important factor is staff training. Employees must understand the correct usage of materials. Mismanagement can lead to errors and increased expenses. Incorporating better practices into daily operations can significantly improve efficiency. Regular workshops can make a difference. Encouraging feedback from staff can reveal areas for improvement.
Sometimes, organizations overlook the importance of planning. Without a solid strategy, resources may be wasted. Staying adaptable to changes in demand can help in adjusting production schedules. Reviewing processes periodically can highlight inefficiencies. Small changes can lead to substantial savings. Balancing quality and cost is always a challenge but vital for success.
Risks and Mitigation in Handling Pharmaceutical Materials in Supply Chain
Handling pharmaceutical materials in the supply chain presents unique challenges. Risks are often tied to environmental factors, contamination, and human error. Temperature changes can alter the efficacy of sensitive compounds. Even minor temperature fluctuations during transport can lead to significant losses.
Another risk involves improper storage conditions. Warehouses must maintain strict humidity and temperature controls. Failure to do so may compromise product integrity. Training staff on proper protocols is essential but frequently overlooked. Team members may not fully grasp the importance of following guidelines. Regular audits can reveal lapses in compliance and areas for improvement.
Communication is key in mitigating risks. All stakeholders need to share information about product specifications and handling procedures. A clear understanding reduces the likelihood of mistakes. However, gaps often exist, leading to potential hazards. Investing time in training and maintaining open lines of communication is crucial for maintaining safety and efficacy throughout the supply chain.
Pharma Material Management Risks and Mitigation Strategies
Related Posts
-

Unique Uses of Pharmaceutical Chemical Powder in Modern Medicine: Unveiling Real-World Applications
-

Navigating the Future of Best Pharmaceutical Materials in 2025 and How to Stay Ahead
-
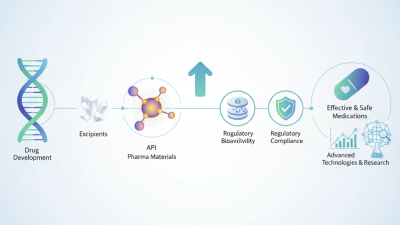
What is Pharma Material and Its Importance in Drug Development
-
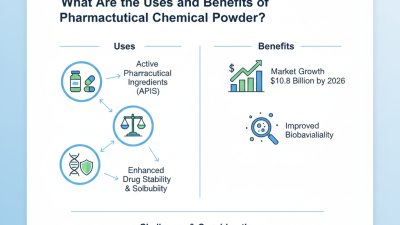
What Are the Uses and Benefits of Pharmaceutical Chemical Powder?
-
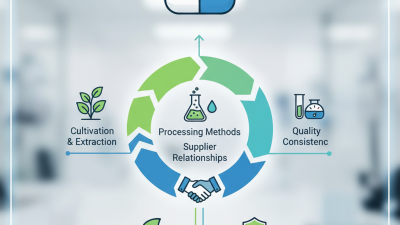
The Best Drug Raw Material Sources for Quality Pharmaceutical Production?
-

Top Pharmaceutical Intermediates You Need to Know About?
